doi: 10.1128/JCM.40.4.1549-1552.2002
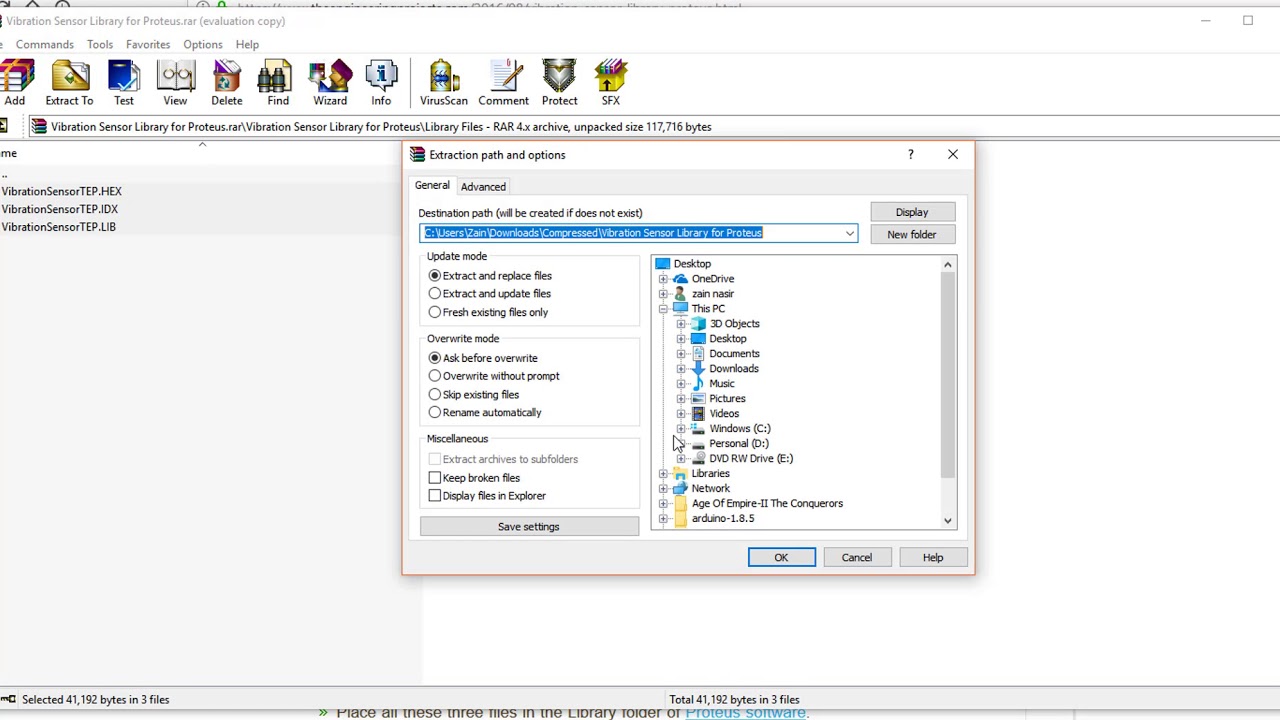
Hello friends, I hope you all are fine and having fun with your lives. Today, I am going to share a list of New Proteus Libraries for Engineering Students.I have shared many Proteus Libraries till now but they all are randomly spread in blog posts so today I thought to post all those Proteus Libraries links in this post. So that engineering students can get benefit from these awesome libraries.
PMID: 11923394
This article has been cited by other articles in PMC.
Abstract
β-Lactamase production was detected in 147 (52%) of 282 consecutive nonduplicate Proteus mirabilis isolates obtained over a 1-year period from the S. Matteo Hospital of Pavia (northern Italy). Seventy isolates (48% of the β-lactamase producers) were found to produce extended-spectrum β-lactamases (ESBLs), identified as PER-1 (first report in this species) and TEM-52 in 52 and 18 isolates, respectively. Analysis of clonal diversity of the ESBL producers suggested different spreading patterns for the two ESBL determinants.
Proteus mirabilis is the second most common cause of urinary tract infections and is also an important cause of nosocomial infections ().
Wild-type strains of P. mirabilis are usually susceptible to ampicillin and other β-lactams, which are among the drugs of choice. However, a progressive increase of β-lactam resistance, mediated by the production of acquired β-lactamases, has occurred in this species (, , , ). Recently, extended-spectrum β-lactamases (ESBLs) active on expanded-spectrum cephalosporins have also started spreading in P. mirabilis, including most frequently TEM-type derivatives (, , , , , , -) but also other enzymes of group 2be (, ).
In this work a survey was carried out on ESBL production by P. mirabilis clinical isolates from an Italian hospital where increased resistance rates to expanded-spectrum cephalosporins, mediated by similar enzymes, were recently observed among other Enterobacteriaceae ().
β-Lactam susceptibility and β-lactamase production in P. mirabilis isolates.
During a 1-year period (February 1997 to January 1998), 282 consecutive nonduplicate P. mirabilis isolates were collected from inpatients at the S. Matteo I.R.C.C.S. Hospital of Pavia (northern Italy). Most isolates (65%) were from urine or other urogenital specimens, while a minority were from respiratory specimens (11%), discharge of infected wounds/ulcers (10%), pus (6.5%), blood (3%), or other sources (4.5%). All isolates were identified using the GNI card of the Vitek system (BioMérieux, Rome, Italy).
Screening by Vitek GNS cards (BioMérieux) revealed that 135 (48%) of the 282 isolates were susceptible to ampicillin and other β-lactams, while the remaining 147 were intermediate or resistant to ampicillin and produced β-lactamase activity detectable by the nitrocefin hydrolysis test (Oxoid-Unipath, Milan, Italy). Susceptibility testing by disk diffusion (15) showed that (i) all the β-lactamase producers were susceptible to cefoxitin, suggesting that production of AmpC-like enzymes was not involved; and (ii) none of them were resistant to amoxicillin-clavulanate but susceptible to cephalothin, suggesting that production of inhibitor-resistant TEMs or oxacillin-type enzymes was also not involved (). Production of ESBL activity was screened by a double-disk synergy test as described previously (), placing the disks at a distance of 23 mm (center to center) from each other. A potentiation of the inhibitory zones of cefotaxime, ceftazidime, cefepime, and/or aztreonam by clavulanate, suggesting the production of ESBL activity, was observed with 70 (48%) of the β-lactamase producers.
Susceptibility testing by a broth macrodilution procedure (14) revealed that the putative ESBL producers exhibited variable susceptibility patterns to cefotaxime (MIC range, 4 to >64 μg/ml), ceftazidime (MIC range, 2 to 32 μg/ml), cefepime (MIC range, 2 to 64 μg/ml), and aztreonam (MIC range, 2 to 32 μg/ml), while the ESBL-negative β-lactamase-producing isolates were always susceptible to the above drugs; for those isolates, MICs were ≤1 μg/ml.
Characterization of ESBLs.
Analytical isoelectric focusing (IEF) of crude extracts of the 70 putative ESBL producers and detection of β-lactamase bands by nitrocefin were carried out as described previously (). The activity against cefotaxime, ceftazidime, cefepime, and aztreonam of the β-lactamase bands separated by IEF was assayed by a substrate overlaying procedure as reported previously (), using a final substrate concentration of 1 μg/ml in the medium overlay and Escherichia coli ATCC 25922 as an indicator strain. Substrate hydrolysis was indicated by the occurrence of bacterial growth above the enzyme bands. IEF detected two β-lactamases of pIs 5.2 and 5.6, respectively, in 52 isolates (in 7 of them the pI 5.2 enzyme appeared to be consistently less abundant than in the other 45) and a single β-lactamase of pI 5.9 in the remaining 18 isolates. In the bioassay, the pI 5.2 and 5.9 enzymes were active on cefotaxime, ceftazidime, cefepime, and aztreonam, while the pI 5.6 enzyme did not show activity with any of the above compounds.
In the 70 ESBL producers the presence of β-lactamase genes was investigated by colony blot hybridization (26). The probes were PCR-generated amplicons containing the entire blaTEM-1 () or blaPER-1 () coding sequence, labeled with 32P using a commercial kit (Rediprime II; Amersham Pharmacia). Results of these experiments showed that all 70 isolates were recognized by the blaTEM probe, while 52 (corresponding to those producing a pI 5.2 enzyme) were also recognized by the blaPER probe (Table (Table1).1).
TABLE 1.
Results of analytical IEF and of molecular characterization of β-lactamase determinants carried by 70 putative ESBL producersd
| No. of isolates | IEF band(s)a | Result of colony hybridization | Enzyme(s)b | MICc (μg/ml) of: | ||||
|---|---|---|---|---|---|---|---|---|
| blaTEM probe | blaPER probe | CTX | CAZ | FEP | ATM | |||
| 52 | pI 5.2e + pI 5.6 | + | + | TEM-2 + PER-1 | 16->64 | 16-32 | 32-64 | 4-32 |
| 18 | pI 5.9 | + | − | TEM-52 | 4-16 | 2-8 | 2-16 | 2 |
aIn the bioassay, the pI 5.2 and 5.9 bands showed activity against cefotaxime, ceftazidime, cefepime, and aztreonam, while the pI 5.6 band did not show activity against any of the above compounds.
bAccording to the sequence of the determinant encoding the mature enzyme.
cCTX, cefotaxime; CAZ, ceftazidime; FEP, cefepime; and ATM, aztreonam. The MICs of ESBL producers were determined by standard broth macrodilution test (14).
dIn vitro susceptibility to expanded-spectrum cephalosporins and aztreonam is also shown.
eThe pI 5.2 enzyme was produced at a lower level in seven of these isolates, as shown by a lower intensity of the band consistently observed in replicated experiments after loading the same amount of crude extracts. Assay of cefotaximase activity in the extracts of three isolates, including two of those producing a higher amount of the enzyme and one of those producing a lower amount (the same isolates selected for sequence analysis, see text), revealed a higher activity in the former isolates (specific activities, 310 ± 35 and 335 ± 40 nmol/min/mg of protein) than in the latter isolate (specific activity, 195 ± 20 nmol/min/mg of protein). No consistent differences, however, were observed between the MICs of CTX, CAZ, FEP, and ATM for isolates producing the enzyme at a higher level and those for isolates producing the enzyme at a lower level.
PCR amplification of blaTEM alleles was carried out with primers TEM/f (5′-GGAAGAGTATGAGTATTCAACAT) and TEM/r (5′-ATATGAGTAAGCTTGGTCTGACAG) and the following parameters: initial denaturation at 94°C for 7 min, denaturation at 94°C for 45 s, annealing at 52°C for 45 s, and elongation at 72°C for 60 s, repeated for 35 cycles; and a final extension at 72°C for 7 min. PCR amplification of blaPER alleles was carried out with primers BLAPER-f (5′-GGGACA(A/G)TC(G/C)(G/T)ATGAATGTCA) and BLAPER-r (5′-GGG(C/T)(G/C)GCTTAGATAGTGCTGAT) as described previously (). PCR was performed using the Expand PCR system (Roche Biochemicals, Mannheim, Germany) under the conditions recommended by the manufacturer, with crude bacterial lysates (obtained by boiling cells in distilled water) as templates. Sequencing of the β-lactamase determinants was performed directly on PCR-generated amplicons on both strands as described previously (). For each gene, the sequenced regions included those encoding the mature protein and part of the signal peptide. Direct sequencing of the blaPER and blaTEM amplicons obtained from three isolates producing the pI 5.2 and 5.6 enzymes, randomly selected (two among those producing a higher amount of the pI 5.2 enzyme and one among those producing a lower amount of the enzyme, all of which were clonally related; see below), always revealed the presence of alleles encoding mature enzymes identical to PER-1 () and TEM-2 (). Direct sequencing of the PCR-amplified blaTEM genes obtained from three isolates producing the pI 5.9 enzyme (selected as representatives of the three circulating clones, see below) revealed, in all cases, the presence of an allele encoding a mature enzyme identical to TEM-52 () (Table (Table11).
Unlike in other reports, where TEM-type enzymes were found to represent the most common ESBLs in P. mirabilis (, ), in this case an “unconventional” class A enzyme was found to be the most prevalent ESBL. PER-1 has recently been reported in Pseudomonas aeruginosa and other gram-negative nonfermenters from northern Italy (, ). Present findings indicate that, in the same area, PER-1 is also emerging in P. mirabilis, a species where this enzyme was not previously reported. The presence of TEM-52 was not surprising since it had already been detected in Klebsiella pneumoniae isolates from the same hospital () and is the most widespread TEM-type ESBL circulating in Enterobacteriaceae in Italy ().
Concerning in vitro susceptibility, the PER-1 producers were always resistant or intermediate to cefotaxime, ceftazidime, and cefepime and, in some cases, also to aztreonam. On the contrary, the TEM-52-producers were intermediate or susceptible to cefotaxime and cefepime and were always susceptible to ceftazidime and aztreonam (Table (Table1),1), a finding that underscores the need for definition of breakpoints for screening ESBL production also in this species. The success of PER-1 could be due, at least in part, to the higher levels of resistance exhibited by the PER-1-producing strain, which also produced TEM-2, and emphasizes the potential clinical relevance that ESBLs other than of the TEM or SHV type could acquire in Enterobacteriaceae.
Clonal relationships and nosocomial distribution of ESBL producers.
The clonal relationships among ESBL producers were investigated by comparing the pulsed-field gel electrophoresis (PFGE) profiles of genomic DNA digested with SfiI. PFGE profiles were analyzed by the Bio-Rad Gene Path Procedure (Bio-Rad Laboratories, Richmond, Calif.) using the no. 5 pathogen group reagent kit. DNA fragments were electrophoresed in 1% agarose gels in 0.5× Tris-borate-EDTA buffer (26) with the Gene Path system (Bio-Rad) at 14°C and 6 V/cm for 20 h, using pulse times ranging from 5 to 50 s. Bacteriophage lambda concatemers (Bio-Rad) were used as DNA size markers. Clonal relationships based on PFGE patterns were interpreted according to the criteria proposed by Tenover et al. (). Profiles identical or different by no more than four bands, indicating clonal relatedness, were observed with all the PER-1 producers (Fig. (Fig.11 and data not shown), suggesting that spreading of this resistance determinant had been most likely contributed by clonal expansion of a strain that had acquired it. On the other hand, profiles different by more than four bands, indicating the presence of three different clonal lineages, were detected among the isolates producing TEM-52 (Fig. (Fig.11 and data not shown). None of the TEM-52-producing clones was apparently related to that producing PER-1 (Fig. (Fig.11).
PFGE patterns of the genomic DNAs of P. mirabilis isolates producing ESBLs, after digestion with SfiI. Lane 1, DNA size standards (in kilobases, on the left); lanes 2 to 4, isolates producing the TEM-52 enzyme representative of the three identified clonal lineages (A, B, and C) (the PFGE patterns of the other 15 TEM-52-producing isolates were identical or related to those shown in the figure); and lanes 5 to 7, isolates producing the PER-1 enzyme; the PFGE profiles of the other 49 PER-1-producing isolates were identical or related to those shown in the figure.
Both the PER-1 and the TEM-52 producers were present since the beginning of the survey and were isolated throughout the survey period. The PER-1-producing strain was initially isolated from patients of a general intensive care unit and, subsequently, also from patients of 15 additional wards (Table (Table2),2), suggesting a remarkable spreading ability. The TEM-52 producers exhibited a more restricted distribution (Table (Table2),2), and a clear relationship was observed between clonality and ward (Table (Table2),2), suggesting that horizontal transfer of the resistance determinant could have played a relevant role in its dissemination.
TABLE 2.
Distribution of ESBL-producing isolates of P. mirabilis in different wards of S. Matteo Hospital, Pavia, Italy
| Ward | No. of isolates | |
|---|---|---|
| PER-1 producinga | TEM-52 producingb | |
| General intensive care unit | 22 (4 LL) | 7 (A) |
| Internal medicine 1 | 1 | NDc |
| Internal medicine 2 | 1 | ND |
| Internal medicine TM | 1 | ND |
| Internal medicine PMd | 5 (1 LL) | 2 (C) |
| Internal medicine 16d | ND | 1 (C) |
| Internal medicine 19d | 2 | 4 (C) |
| Medicine—long-term 1 | 1 | 4 (B) |
| Medicine—long-term 2 | 1 | ND |
| Infectious diseases | 1 | ND |
| Respiratory diseases | 2 | ND |
| General surgery 2 | 2 | ND |
| General surgery 5 | 1 (1 LL) | ND |
| Vascular surgery | 4 (1 LL) | ND |
| Neurosurgery | 7 | ND |
| Orthopedics | 1 | ND |
| Total | 52 | 18 |
aThe PER-1-producing isolates were all clonally related. LL indicates an isolate that produced lower levels of the enzyme in IEF analysis (see text and Table Table11 for more details).
bThe capital letters indicate the different clones.
dThese wards were located at different levels of the same block.
Concluding remarks.
The prevalence of ESBL-producing P. mirabilis isolated from this hospital was notably higher than that observed in other settings (). The remarkable epidemiological variability of this phenomenon, which can acquire a notable impact, suggests that screening for ESBL production should be routinely considered also for this species.
Acknowledgments
This work was supported in part by a grant from “F.A.R., Fondo di Ateneo per la Ricerca,” University of Pavia, to L.P.
Thanks are due to Emanuela Dell'Amico for an excellent technical contribution in the sequencing work.
REFERENCES
1. Ambler, R. P., and G. K. Scott. 1978. Partial amino acid sequence of penicillinase coded by Escherichia coli plasmid R6K. Proc. Natl. Acad. Sci. USA75:3732-3736. [PMC free article] [PubMed] [Google Scholar]
2. Bauernfeind, A., I. Stemplinger, R. Jungwirth, P. Mangold, S. Amann, E. Akalin, O. Ang, C. Bal, and J. M. Casellas. 1996. Characterization of β-lactamase gene blaPER-2, which encodes an extended-spectrum class A β-lactamase. Antimicrob. Agents Chemother.40:616-620. [PMC free article] [PubMed] [Google Scholar]
3. Bonnet, R., C. De Champs, D., Sirot, C. Chanal, R. Labia, and J. Sirot. 1999. Diversity of TEM mutants in Proteus mirabilis. Antimicrob. Agents Chemother.43:2671-2677. [PMC free article] [PubMed] [Google Scholar]
4. Bonnet, R., J. L. Sampaio, R. Labia, C. De Champs, D. Sirot, C. Chanal, and J. Sirot. 2000. A novel CTX-M β-lactamase (CTX-M-8) in cefotaxime-resistant Enterobacteriaceae isolated in Brazil. Antimicrob. Agents Chemother.44:1936-1942. [PMC free article] [PubMed] [Google Scholar]
5. Chanal, C., R. Bonnet, C. De Champs, D. Sirot, R. Labia, and J. Sirot. 2000. Prevalence of β-lactamases among 1,072 clinical strains of Proteus mirabilis: a 2-year survey in a French hospital. Antimicrob. Agents Chemother.44:1930-1935. [PMC free article] [PubMed] [Google Scholar]
6. De Champs, C., C. Monne, R. Bonnet, W. Sougakoff, D. Sirot, C. Chanal, and J. Sirot. 2001. New TEM variant (TEM-92) produced by Proteus mirabilis and Providencia stuartii isolates. Antimicrob. Agents Chemother.45:1278-1280. [PMC free article] [PubMed] [Google Scholar]
7. Fluit, A. C., M. E. Jones, F. J. Schmitz, J. Acar, R. Gupta, and J. Verhoef. 2000. Antimicrobial resistance among urinary tract infection (UTI) isolates in Europe: results from the SENTRY Antimicrobial Surveillance Program 1997. Antonie Leeuwenhoek77:147-152. [PubMed] [Google Scholar]
8. Jarlier, V., M. H. Nicolas, G. Fournier, and A. Philippon. 1988. Extended broad-spectrum β-lactamases conferring transferable resistance to newer beta-lactam agents in Enterobacteriaceae: hospital prevalence and susceptibility patterns. Rev. Infect. Dis.10:867-878. [PubMed] [Google Scholar]
9. Jones, R. N., K. C. Kugler, M. A. Pfaller, and P. L. Winokur. 1999. Characteristics of pathogens causing urinary tract infections in hospitals in North America: results from the SENTRY Antimicrobial Surveillance Program, 1997. Diagn. Microbiol. Infect. Dis.35:55-63. [PubMed] [Google Scholar]
10. Liu, P. Y. F., D. Gur, L. M. C. Hall, and D. M. Livermore. 1992. Survey of the prevalence of β-lactamase amongst 1000 gram-negative bacilli isolated consecutively at the Royal London Hospital. J. Antimicrob. Chemother.30:429-447. [PubMed] [Google Scholar]
11. Luzzaro, F., M. Perilli, G. Amicosante, G. Lombardi, R. Belloni, A. Zollo, C. Bianchi, and A. Toniolo. 2001. Properties of multidrug-resistant, ESBL-producing Proteus mirabilis isolates and possible role of β-lactam/β-lactamase inhibitor combinations. Int. J. Antimicrob. Agents17:131-135. [PubMed] [Google Scholar]
12. Luzzaro, F., E. Mantengoli, M. Perilli, G. Lombardi, V. Orlandi, A. Orsatti, G. Amicosante, G. M. Rossolini, and A. Toniolo. 2001. Dynamics of a nosocomial outbreak of multidrug-resistant Pseudomonas aeruginosa producing the PER-1 extended-spectrum β-lactamase. J. Clin. Microbiol.39:1865-1870. [PMC free article] [PubMed] [Google Scholar]
13. Mariotte, S., P. Nordmann, and M. H. Nicolas. 1994. Extended-spectrum β-lactamase in Proteus mirabilis. J. Antimicrob. Chemother.33:925-935. [PubMed] [Google Scholar]
14. National Committee for Clinical Laboratory Standards. 2000. Methods for dilution antimicrobial susceptibility tests for bacteria that grow aerobically. Approved standard M7-A5. NCCLS, Wayne, Pa.
15. National Committee for Clinical Laboratory Standards. 2000. Performance standards for antimicrobial disk susceptibility tests. Approved standard M2-A7. NCCLS, Wayne, Pa.
16. Nordmann, P., and T. Naas. 1994. Sequence analysis of PER-1 extended-spectrum β-lactamase from Pseudomonas aeruginosa and comparison with class A β-lactamases. Antimicrob. Agents Chemother.38:104-114. [PMC free article] [PubMed] [Google Scholar]
17. Pagani, L., M. Perilli, R. Migliavacca, F. Luzzaro, and G. Amicosante. 2000. Extended-spectrum TEM- and SHV-type β-lactamase-producing Klebsiella pneumoniae strains causing outbreaks in intensive care units in Italy. Eur. J. Clin. Microbiol. Infect. Dis.19:765-772. [PubMed] [Google Scholar]
18. Pagani, L., F. Luzzaro, P. Ronza, A. Rossi, P. Micheletti, F. Porta, and E. Romero. 1994. Outbreak of extended-spectrum β-lactamase producing Serratia marcescens in an intensive care unit. FEMS Immunol. Med. Microbiol.10:39-46. [PubMed] [Google Scholar]
19. Palzkill, T., K. S. Thomson, C. C. Sanders, E. S. Moland, W. Huang, and T. W. Milligan. 1995. New variant of TEM-10 β-lactamase gene produced by a clinical isolates of Proteus mirabilis. Antimicrob. Agents Chemother.39:1199-1200. [PMC free article] [PubMed] [Google Scholar]
20. Pereira, M., M. Perilli, E. Mantengoli, F. Luzzaro, A. Toniolo, G. M. Rossolini, and G. Amicosante. 2000. PER-1 extended-spectrum beta-lactamase production in an Alcaligenes faecalis clinical isolate resistant to expanded-spectrum cephalosporins and monobactams from a hospital in Northern Italy. Microb. Drug Resist.6:85-90. [PubMed] [Google Scholar]
21. Perilli, M., B. Segatore, M. R. de Massis, M. L. Riccio, C. Bianchi, A. Zollo, G. M. Rossolini, and G. Amicosante. 2000. TEM-72, a new extended-spectrum β-lactamase detected in Proteus mirabilis and Morganella morganii in Italy. Antimicrob. Agents Chemother.44:2537-2539. [PMC free article] [PubMed] [Google Scholar]
22. Perilli, M., E. Dell'Amico, B. Segatore, M. R. de Massis, C. Bianchi, F. Luzzaro, G. M. Rossolini, A. Toniolo, G. Nicoletti, and G. Amicosante. 2002. Molecular characterization of extended-spectrum β-lactamases produced by nosocomial isolates of Enterobacteriaceae from an Italian nationwide survey. J. Clin. Microbiol.40:611-614. [PMC free article] [PubMed] [Google Scholar]
23. Pitout, J. D. D., K. S. Thomson, N. D. Hanson, A. F. Ehrhardt, E. S. Moland, and C. C. Sanders. 1998. β-Lactamases responsible for resistance to expanded-spectrum cephalosporins in Klebsiella pneumoniae, Escherichia coli, and Proteus mirabilis isolates recovered in South Africa. Antimicrob. Agents Chemother.42:1350-1354. [PMC free article] [PubMed] [Google Scholar]
24. Poyart, C., P. Mugnier, G. Quesne, P. Berche, and P. Trieu-Cuot. 1998. A novel extended-spectrum TEM-type β-lactamase (TEM-52) associated with decreased susceptibility to moxalactam in Klebsiella pneumoniae. Antimicrob. Agents Chemother.42:108-113. [PMC free article] [PubMed] [Google Scholar]
25. Rozalski, A., Z. Sidorezyk, and K. Kotelko. 1997. Potential virulence factors of Proteus bacilli. Microbiol. Mol. Biol. Rev.61:65-89. [PMC free article] [PubMed] [Google Scholar]
26. Sambrook, J., and D. W. Russel. 2001. Molecular cloning, a laboratory manual, 3rd ed. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, N.Y.
27. Sutcliffe, J. G. 1978. Nucleotide sequence of the ampicillin resistance gene of Escherichia coli plasmid pBR322. Proc. Natl. Acad. Sci. USA75:3737-3741. [PMC free article] [PubMed] [Google Scholar]
28. Tenover, F. C., R. D. Arbeit, R. V. Goering, P. A. Mickelsen, B. E. Murray, D. H. Persing, and B. Swaminathan. 1995. Interpreting chromosomal DNA restriction patterns produced by pulsed-field gel electrophoresis: criteria for bacterial strain typing. J. Clin. Microbiol.33:2233-2239. [PMC free article] [PubMed] [Google Scholar]
Articles from Journal of Clinical Microbiology are provided here courtesy of American Society for Microbiology (ASM)
